“Atom” comes from the ancient Greek for “indivisible”, a very optimistic assumption that nothing smaller exists. As summed up in this strange nursery rhyme, there is always something underneath: “Big fleas have small fleas, andlay on their back to bite them and small fleas have smaller fleas and so on.”
100 years ago, Neils Bohr received the Nobel Prize for his atomic model, which is still taught in schools today. Scientists have had all these years to look for the “lesser fleas” and come up with more theories about how our universe is built and reacts all around us. Let’s start at the beginning with the idea that everything is made of something smaller.
The first ideas of the atom came from ancient philosophers like Democritus around 400 BC. Millennia before his time, the idea that there are tiny, invisible spheres that make up everything in the world was, to most people: scary, poppy, and insane. Yes, some would say it still is all of those things, but in the 19th century the facts began to become indisputable.
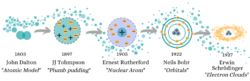
John Dalton was the first to use a “atomic theory‘ in 1803. So he told the world; 1) an atom was the smallest unit of matter, 2) each element had its own atom (distinguished by its weight), and 3) that when atoms interacted with each other, compounds were formed. Right on most counts, Dalton revealed a great new area of physics, but was slightly ahead of himself in using Democritus’ naming of “atoms” as it was soon discovered that these “indivisibles” were actually made up of other particles.
This is where the electron enters! Crazy little whizzing chaos balls. 90 years after Dalton, the idea of the atom was widespread when JJ Thompson pushed the boundaries of atoms as the most basic particle. With this revelation of a tiny negatively charged particle that resides inside the atom, he had to remodel what an atom is and came up with “The plum pudding model“. This found electrons swimming in a sea of positive charge inside the atom (much like raisins hide in a lead pudding).
The beginning of the 20th century brings scientists full of ideas for the new era. One such man was Rutherford, who in 1905 cut open the plum pudding and pulled out all the raisins. He proved that the atom was separated into compartments, a positive nucleus surrounded by empty space containing electrons. (Me and hungry physics students around the world will forever be grateful to him for a model of the atom not named after cake.)
Here Niels Bohr took the stage as Rutherford’s pupil. His 1922 Nobel Prize was for the discovery that the atom was even more organized than in Rutherford’s model. Electrons did not move freely, but were contained to set orbits around the nucleus between which they could jump when energy was applied to them, or fall off through the use of energy. This model makes organized people happy – such a shame that quantum mechanics was lurking just around the corner to mess things up.
Less than five years after this Nobel Prize, Erwin Schrödinger revealed “wave theory“. Instead of orbiting around the nucleus like tiny planets, the particles become waves that fill 3D space. However, it gets really weird when we look at electrons, they behave like particles. So Schrödinger mapped where he found electrons to create a new model of electron clouds.
In 1927, just a year after Schrödinger’s revelation, Heisenberg made this little world even stranger. He tells us that not only does an electron stop behaving like a wave when we look at it, but also that we cannot know an electron’s velocity at the same time as we know its location. We can’t watch it move and Look how fast it goes, we only see the still image. Conversely, if we know how fast it’s going, we have no idea where it is. Appropriately, that’s what it’s called “Uncertainty Principle.”
All of this seems to get a little skipped in high school physics if we jump straight to the point neutron Discovery that did not take place until 1931. Even more from here subatomic particles, as they are known today, from electrons to cottage cheese and last bosons. The atom that was supposed to be the smallest unit of matter has been split into nearly 20 particles, and there are still more to discover.
This area of physics has advanced our understanding of how things interact on a universal scale explain fireworksto shoot subatomic particles at biological proteins discover their formsand the current attempt to rebuild nuclear fusion for sustainable energy. All of these advances are based on our understanding of the atom. While it sounds gross, by observing the “lesser fleas” we can find even smaller fleas and use that knowledge to improve the world around us.